How to calculate the cooling capacity of a chiller. Chillers provide chilled water which is then used to provide air conditioning within buildings. The amount of cooling they produce varies and it’s important to know how much cooling a chiller is producing or is able to produce. There is a video tutorial at the bottom of the page also.
Firstly to perform this calculation we need to know a few things.
- The volume flow rate of water into the evaporator
- The inlet and outlet chilled water temperature
We then need to lookup the properties of water for the following
- The density of the water at the average temperature (inlet temp + outlet temp)/2
- The Specific Heat Capacity of the chilled water at the average temperature (inlet temp + outlet temp)/2
A recommended website to look these properties up is: PeaceSoftware.de
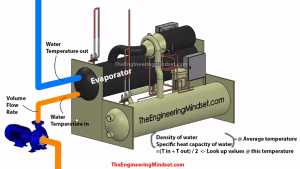
Let look at how to calculate the cooling capacity. We’ll first look at how to calculate in metric units and then imperial.
Metric units:
The water flow rate of chilled water into the evaporator is 0.0995m3/s, the inlet temperature is 12*c and the outlet temperature is 6*c. This means the average temperature is 9*c so we lookup the water properties at this temperature to find the density of 999.78kg/m3 and a specific heat capacity of 4.19kJ/kg/K.
Using the energy equation of Q = ṁ x Cp x ΔT we can calculate the cooling capacity.
Q = (999.78kg/m3 x 0.0995m3/s) x 4.19kJ/kg/K x ((12*c+273.15K) – (6*c+273.15K))
We add 273.15K to the celcius to convert it to units of Kelvin. The Specific heat capacity (Cp) is measured in units of kJ per kg per Kelvin.
This gives us a final answer of Q = 2,500kW of cooling. Full calculations are shown below.

Now lets look at how to calculate the cooling capacity of a chiller in imperial units
Imperial units:
The flow rate of chilled water into the evaporator is measured as 12,649ft3/h and the chilled water inlet temperature is 53.6*F the outlet temperature is 42.8*F. The average temperature is 48.2*F so we need to calculate the water properties at this temperature.
A good website for this is peacesoftware.de the although we will need to convert the units to imperial so for that we will use Specific heat capacity and density of water
This will give us a specifi heat capacity of 1.0007643BTU/lb.F and density of 62.414lb/Ft3
Using the energy equation of Q = ṁ x Cp x ΔT we can calculate the cooling capacity.
Q = (16,649FT3/h x 62.414lb/ft3) x 1.0007643BTU/lb.F x (53.6F – 42.8F)
Giving us a cooling capacity of 8,533,364BTU/h. see full calculations below.




















Hi Paul,
Good day. Need your advice on heat recovery system from water chiller to heat water in building for hot water supply. Requirement parameters are:
1) heat circulating water from ambient inlet 27 degC to supply outlet 60 degC;
2) current chiller design 35 kW;
3) chilled water supply inlet 14.8 degC and return outlet 8 degC.
Please advise with justification/calculations if this system chiller is able to perform to its requirements.
(Hot water supply to roof storage tanks usage 12 cu.metres per day)
Also, the heat loss on a 50mm diameter with 25mm insulation over a distance from chiller to roof storage tanks is est. 100m in length. What’s the possible heat loss?
Appreciate for prompt reply Paul. Thank you & cheers.
Best regards,
Tony
hi
good day,
please calculate pressure or temp for compression refrigeration cycle 7kw?(r-134a)
later than i know 7kw
i want to know how i calculate comp cond evap.
Hi,
Like to ask. For instance a chiller is connected to 4 AHU. IF I manage to use analytics to understand my actual demands and use it to reduce the chill water flow rate and optimise the delta temperature of chill water inlet and outlet at AHU level, and save an average of 30% energy saving using btu meters measure at the 4 ahu chill water.
Does saving this 30 % energy of chill water Meanthat I could be saving 30% energy or more of the whole hvac system (chiller, compressor, pump)? Reason for having this assumption is if my chill water temperature and flow is optimise, my chiller will work lesser to produce the output which is chill water.
Am I right in this assumption?
For your guidance.
Hello dear Mr. Paul have a nice day.
Hii I wish to know how much water temperature outlet and how inlet in chiller
Hello sir,
i have a chiller 260 tr if chiller running 12 hours per days , it is possible may we count line loss kwh to kwah, and reply for minimum line loss.
Sir how to confirm chiller how much tr
Formula
Hi Sir,
Can u suggest me suitable flow meter to measure the flow rate of Air Cooled 240 TR Chiller.
Line size is 6 inch
how to calculate transformation lose of ammonia chiller
How to select pipe size for chiller line if pump capacity known..
what if the water is 25% glycol based? how to identify the density & Cp in this condition? do you have any website where we can verify it? like you mentioned for the water ‘peacesoftware.de’
I am trying to figure out my chillers cooling capacity at variant outlet temperatures, while maintaining the same ambient temperature. For example, I have a low temperature chiller that provides 5RT (60,000 Btu)at a leaving temperature of 5°F while at 95°F ambient temperature
how can I calculate its cooling capacity at 10°F, 20°F, 30°F 40°F outlet temperatures.
50kw liquid chiller unit:
For above chiller how much air flow required for gud efficiency. As well as how to calculate the aluminum finned type heat exchanger. Please suggest use.
So I’m on a chilled water system in my condo. The water originates from a well in the ground and returns to after running through the air handlers. Does the water run around the coils? Do you have a diagram. Is there a wat to clean the coils that the well water runs through
So I’m on a chilled water system in my condo. The water originates from a well in the ground and returns to the well after running through the air handlers. Does the water run around the coils? Do you have a diagram. Is there a way to clean the coils that the well water runs through
Hii paul, thanks for explaining in detail, can you please tell us the working of economizer in a chiller unit.
Hi, is there a typo in the metric calculation of BTU/hr? it shows 8xx,xxx.xx instead of 8 million xxx. Or am I missing something?
Hi I am Vishal my question if 150TR compreesor how much ampere load it takes
Dear Vishal,
Usually it depends on the motor that we include in the system, If its a 125 hp motor then the power consumption would be 125 x 0.7 = 87.5 amps (considering ~70% load).
Best Regards,
AJAY K
250 amps
[…] We previously looked at how to calculate the cooling capacity of a chiller, and for this you would need to know the flow rate of water through the chiller. If you haven’t seen that then click here. […]
Hi Paul,
I’m planning to set-up a business in agricultural sector. My client wanted me to provide chilled water that will run to their hydroponic system. Actually they wanted me to use their existing 3-hp compressor, recoil it and put it inside a pipe where water will pass through. We are expecting water will cool down as it leaves the unit and goes directly to their hydroponic set-up. I’m finding difficulty in getting the cooling surface area suitable for 3-hp compressor. Do you have a table where i can see required cooling surface area (of copper tubes or finned tubes) for any compressor capacity? I appreciate if you can help me on this.
thank you
I’m referring to evaporator tubes surface area suitable for 3-hp compressor. The idea is to modify the evaporator, coil it inside a pipe where water will pass through.
not really sure if this will work or will give us satisfactory chilled water to irrigate the hydroponic plants.
dear sir,
hope you good time
a space needs a cooling unit of 10 tr ,if hrv of 50% capacity is going to be used ,what should be the capacity of the cooling unit.
If i have a water tank of 15 m3 , then i want to cool the tank to 5 c , whats the capacity should be of my chiller ??
If i have a water tank of 15 m3 , then i want to cool the tank to 5 c , whats the capacity should be of my chiller ??
/.;L
Sir,
Chiller standard evaporator how much rate of flow GPM or Cu.m /Sec can consider to calculate chiller capacity?
Hi sir, thank you for calculations
how we can calculate water flow rate by RT
Hi,Paul, Can you guide me to calculate COP of a Direct Fired Absorption chiller after calculating Chiller capacity as per formula given at your web.
Will it be depend on inlet N.Gas consumption + Electrical Load.
Out put capacity/input energy.
Best Regards.
M.Kaleem Farooqi.
T1=-39 C,T2=-41 ——-T average=-39.5 what is the density if you have a table
Regards
Hi,
Im trying to calculate BTU for chiller that has to do following thing:
Cool 200 tons of sea water from 22C to 2C in 24 hours. There are 2 options:
A) 22C inlet and 2C outlet temp with water flow 200 ton/day
B) Big water tank and recirculating water.
Would you be so kind to help me with a rough estimate.
Thank you,
Best regards
Hi Mike,
You need approximately 225 tr Chiller or 800kw Chiller. A normal 20ft Chiller with 225Tr capacity can do.
HI I HAVE A ammonia chiller on this chiller refrigerant is ammonia, air is cooled by ammonia in evaporator. my doubht is i want to calculate chiller TR, what parameters to be taken for caliculate the TR and one more chiller is there which is cooled nitrogen same like air.so how caliculate the both chillers TR.
great to read. so informative.
question:
what limitation we have for the water flow rate?
the higher flow rate will lead to higher cooling capacity, but what constrains will have at high water flow?
thanks
Hello Mr. Paul Evans-
I went through your website and the YouTube tutorial and found a very good explanation for determining cooling load. I’m stuck at this point in my case. We are currently running a 2 TR chiller (with R 407 refrigerant). We want to find the exact size of a chiller for our machine. water inlet temperature: 15 °C The water out temperature is 65 degrees Celsius, and the water mass flow rate is 20 LPM. I put these values in the formula Q = m*cp*(T1-T2). My answer is approximately 21 tonnes. but it is not correct in practical situations. Please help me.