Density Explained. Each material has a different density, the difference depends on how the materials particles are packed together. If the particles of a material are stored very neatly, and closer together, then more particles will be able to fit into the volume. Since particles have mass, the more particles you can fit into the volume, the more the material will weigh. Scroll to the bottom to watch the YouTube video tutorial
Density is just a measurement of how much mass is in a given volume. We measure density in the SI units of kg/m3 (kilograms per cubic meter) usually density is listed as the Greek symbol Rho ρ.
Good news! we have already listed the typical density of common metals, just click here to view the table. (opens in a new tab).
If you find this page useful, please like, share and comment.
Why do we need to know the density of materials
Knowing the density of materials is very useful. Designers will estimate the weight of a product by multiplying the volume by the density of a material, this is very useful if a product had to be designed within a certain weight limit. You can also identify a material by measuring its volume and mass.
How to calculate density
Density is calculated simply by dividing the mass (m) of an object by its volume (V).
ρ = m / V
Mass has the SI units of kilogram (kg) and volume has the SI units of cubic meter (m3)
ρ = kg / m3
Example
You have a large solid metal block but are unsure if it is made from Aluminium or Stainless steel. You measure it and find it has a volume of 2m3 and a total mass of 5400kg.
You calculate it’s density using the formula
ρ = m / V
ρ = Density = kg/m3
m = Mass = kg
V = Volume = m3
So drop the numbers of your measurements into the equation:
ρ = 5400kg ÷ 2m3
and the answer is
ρ = 2700kg/m3
Aluminium has a density of approx 2705kg/m3 and Stainless Steel has a density of approx 7982kg/m3. So the block must be made from Aluminium. Click here to find the density of other common metals.
Important – Density of fluids
The density of solids remains almost constant, however, the density of a fluid will vary depending on pressure and temperature.
If a fluid is heated, then its mass will remain constant but the volume will increase, this means that the density will decrease.
If a fluid is compressed, the mass remains the same but the volume decreases so the density will increase.

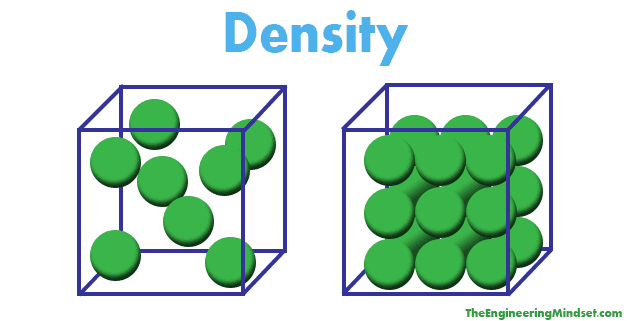
















[…] see how density of materials is calculated, click here. If you find this article useful then please like, share and […]
[…] If you don’t understand what “density” means, then read the tutorial first, click here. […]
hi
I think is like a measure of mass per unit volume. Density is a measure of mass per volume. An object made from a relatively dense material (such as iron) will have less volume than an object of equal mass from a less dense substance (such as water).
what i learned is that the measure of the mass of an object per unit volume how a pese of wood can float but a small rock sinks that is because of how it is shaped
Hi Paul. My name is Gerard. I’m an HVAC Technician in the New York City metropolitan area. I enjoy and appreciate the knowledge you share, the simplicity of your animations, and the engineering data. My question doesn’t fit this forum but I didn’t notice any other location to post this question so please excuse me. Do you have any videos and explanations of small, large, air cooled and water cooled rotary screw chillers? If so, where can I find them?
Hey Gerard, we have a video showing the basics of how each type of compressor works here: https://youtu.be/7Bah__spkTY then videos on water cooled chillers here: https://youtu.be/0rzQhSXVq60 and air cooled here: https://youtu.be/Ic5a9E2ykjo
Great paul, but is there any way to calculate the density of liquid mixtures
I think is like a measure of mass per unit volume. Density is a measure of mass per volume. An object made from a relatively dense material (such as iron) will have less volume than an object of equal mass from a less dense substance (such as water).
Density is not only important in the engineering but also in occupational health and safety. Carbon monoxide and air have different density. by knowing this fact, we will be able to take the right actions when there is a carbon monoxide leak.
Hi Paul,
Your analysis is simple and easy to understand. I just feel to add some analysis further to density change due to heating. Interestingly, while heating water in a container, the water molecules circulate inside the container, due to changes in the density values of water. The density of water will be decreased due to heating, these leayer of water molecules start moving upwards leading to relatively denser (colder water molecules) occupy the vacant space..that is at low level in the container, which is continuously heated. Again, these colder molecules get heated and density is reduced causing them to move upwards and other colder molecules reach this space… this process continues. Finally at boiling point of water, that is around 100 Dec Celcius, the phase of water to changes to steam (vapour). These vapour form of water which are seemingly in the form of bubbles (very low density) will move briskly towards upwards and escape from the surface of liquid to outside…of course there is a role of vapour pressure at this stage, to influence the formation of water bubbles as well…
Great paul