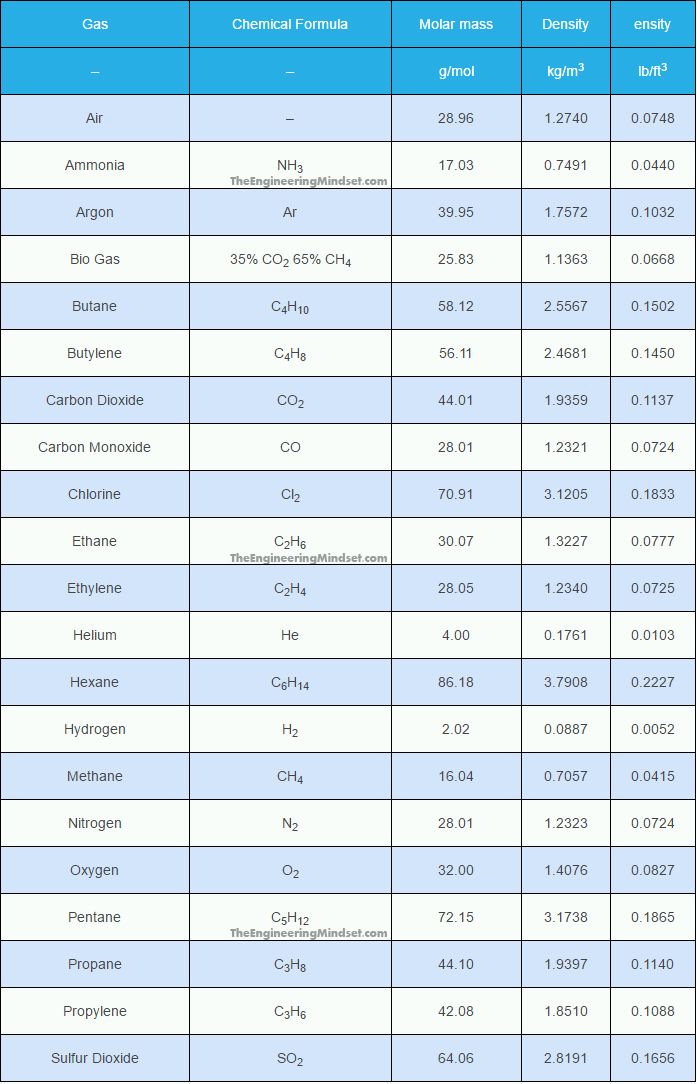
The density of gases have been listed below in alphabetical order in the units of both metric and imperial. The chemical formula as well as molar mass has also been listed.
below the table is an image version for offline viewing
| Gas | Chemical Formula | Molar mass | Density | Density |
|---|---|---|---|---|
| – | – | g/mol | kg/m3 | lb/ft3 |
| Air | – | 28.96 | 1.2740 | 0.0748 |
| Ammonia | NH3 | 17.03 | 0.7491 | 0.0440 |
| Argon | Ar | 39.95 | 1.7572 | 0.1032 |
| Bio Gas | 35% CO2 65% CH4 | 25.83 | 1.1363 | 0.0668 |
| Butane | C4H10 | 58.12 | 2.5567 | 0.1502 |
| Butylene | C4H8 | 56.11 | 2.4681 | 0.1450 |
| Carbon Dioxide | CO2 | 44.01 | 1.9359 | 0.1137 |
| Carbon Monoxide | CO | 28.01 | 1.2321 | 0.0724 |
| Chlorine | Cl2 | 70.91 | 3.1205 | 0.1833 |
| Ethane | C2H6 | 30.07 | 1.3227 | 0.0777 |
| Ethylene | C2H4 | 28.05 | 1.2340 | 0.0725 |
| Helium | He | 4.00 | 0.1761 | 0.0103 |
| Hexane | C6H14 | 86.18 | 3.7908 | 0.2227 |
| Hydrogen | H2 | 2.02 | 0.0887 | 0.0052 |
| Methane | CH4 | 16.04 | 0.7057 | 0.0415 |
| Nitrogen | N2 | 28.01 | 1.2323 | 0.0724 |
| Oxygen | O2 | 32.00 | 1.4076 | 0.0827 |
| Pentane | C5H12 | 72.15 | 3.1738 | 0.1865 |
| Propane | C3H8 | 44.10 | 1.9397 | 0.1140 |
| Propylene | C3H6 | 42.08 | 1.8510 | 0.1088 |
| Sulfur Dioxide | SO2 | 64.06 | 2.8191 | 0.1656 |

















I know only molecular formula and molecular weight of compound, is possible to calculate vapor density? If any one have solution, please put here
hello paul. I have a question here. if I have the ACH value of the room, then I can get the CFM from it right? Then from CFM I can get mass flow rate (m dot) from CFM by multiplying it with the density of air, right? then I can get the cooling load for a room by multiplying m dot with delta h (enthalpy) right?
thank you in advance!
For low pressures, the ideal gas law allows you to calculate what you are looking for
PV=nRT
Hi,
How to calculate a container having 500kg of liquidated argon gas into cylinder bottle having a temp of 20 to 30 degree cellius.
Hi
I need for the density of the exhaust gases from a Diesel fueled gas turbine,