Put simply Electricity is just the flow of free electrons from one atom to another.
Everything in our universe, including you, is made up of atoms. At the very centre of an atom is a group, known as the nucleus, which is made up of protons and/or neutrons. The number of protons and neutrons in each nucleus is what makes materials, or elements, different.
The protons are positively charged, the neutrons have no charge and the electrons are negatively charged. The electrons have the same quantity of charge as the proton, except in the opposite polarity.
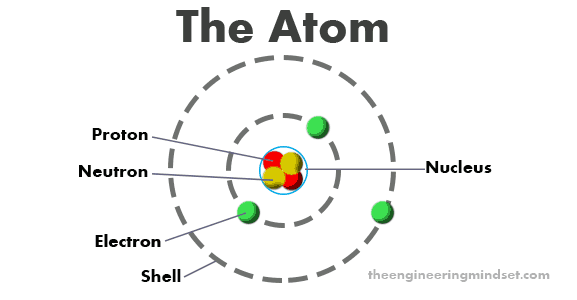
The electrons orbit the nucleus in different paths, known as shells or energy levels. Each shell can hold a maximum number of electrons, the first shell has a maximum of two electrons, the second shell can hold up to eight and the third can hold up to 32 etc.
The outermost shell is known as the valance shell, this orbit is the easiest for electrons to be knocked out of or join. The shell closest to the nucleus is the most difficult for electrons to move to or from.
It is easier for some materials than others for electrons to be knocked out of or join the valance shell. Materials such as copper and aluminium can easily do this but wood and ceramics find it very difficult. Those that find it easy are called conductors and those that find it difficult are called insulators.
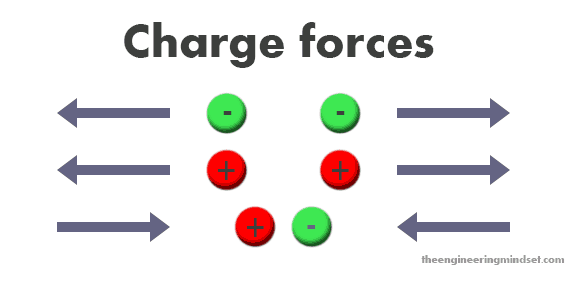
The charge of the particles and their behaviour to each other is a very important part of generating electricity. Opposite charges attract each other and alike charges will repel each other, you may have seen this when moving two magnets near one another.
When two electrons approach each other, they will try to force each other away because they have alike charges, however they will be attracted by a protons positive charge. This attraction and repulsion helps the atom stay together but we can also manipulate this to enable us to generate electricity.
The atoms in a copper wire can easily lose or gain electrons from it’s valence shell. We can eject an electron from the valence shell by forcing it out with another electron or pull it out by attracting it with a proton. When an electron leaves the valence shell it is known as a “free electron” which can move around in any direction within the space between atoms. If we connect the copper wire to a battery and make a circuit, then we can force electrons to be ejected from the valence shell where they will flow in the same direction, and thus we have electricity.
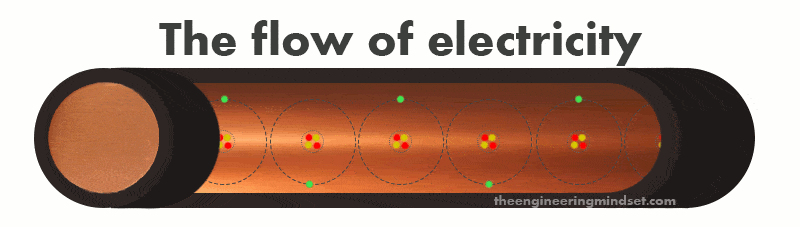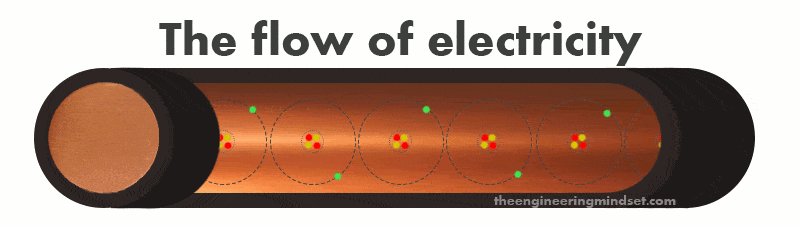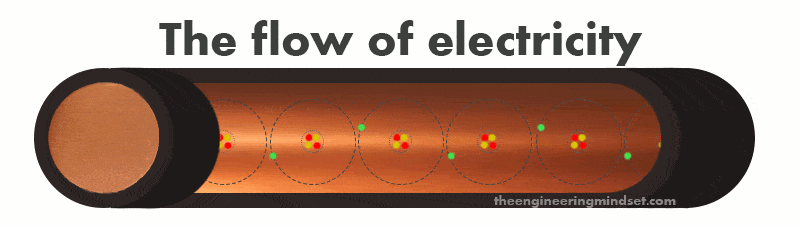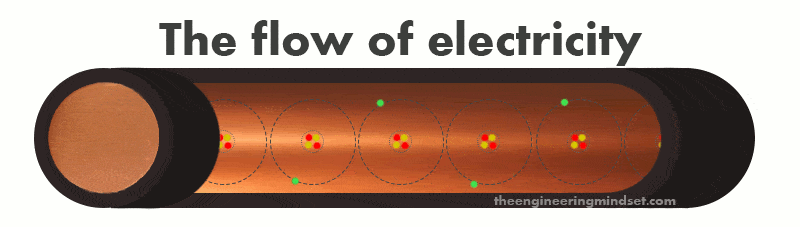
The electrons will be forced to move because a battery has an excess of electrons on the negative terminal and an absence of electrons on the positive terminal. Once a circuit is made, the negative terminal will try to force electrons through the atoms of the copper wire so that they can reach the positive terminal.
Summary
- Electricity is the flow of electrons between atoms
- Atoms which can easily move electrons are called conductors
- Atoms which can’t easily move atoms are called insulators
- Particles with alike charges repel each other
- Particles with opposing charges attract each other
- We manipulate charge attraction and repulsion to generate electricity
- Electricity, or electrons, flow from negative to positive terminals

















[…] or electricity, it come in many different forms such as Kinetic, Potential, Thermal, Chemical, Electrical, Electromagnetic, Sound, Nuclear […]
[…] the opposition to the flow of electrons in a circuit through a conductor. If you need to refresh on how electricity works, click here to read the tutorial […]
Thank you very much !
I think a small error has crept in here
“Atoms which can’t easily move atoms are called insulators”
I noticed that as well, in the summary section, 3rd bullet, should be corrected as: [Atoms which can’t easily move “electrons” are called insulators]
thanks, bro so helpful (●’◡’●)
This really helped me learn a lot of new thigs! I never knew anything about electrons, protons, neutrons and more!!! who knew atoms were a thing!! hehe!